Background of Lipids
-contain carbon, hydrogen and oxygen.
-insoluble in water.
-soluble in organic solvents (e.g. alcohols such as ethanol)
-the main lipids are triglycerides (fats/oils), phospholipids and waxes.
-insoluble in water.
-soluble in organic solvents (e.g. alcohols such as ethanol)
-the main lipids are triglycerides (fats/oils), phospholipids and waxes.
Function of Lipids
The main role of lipids is in plasma membranes
-they contribute to the flexibility of the membranes.
-they aid the transfer of lipid soluble substances across them.
Other Roles
-an energy source (when oxidised, lipids provide twice the amount of energy than carbohydrates).
-waterproofing (lipids are insoluble in water).
-insulation (fats are slow conductors of heat so help to retain body heat when under skin).
-protection (fats are stored around delicate organs to cushion them).
-they contribute to the flexibility of the membranes.
-they aid the transfer of lipid soluble substances across them.
Other Roles
-an energy source (when oxidised, lipids provide twice the amount of energy than carbohydrates).
-waterproofing (lipids are insoluble in water).
-insulation (fats are slow conductors of heat so help to retain body heat when under skin).
-protection (fats are stored around delicate organs to cushion them).
Triglycerides
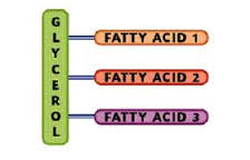
-ester bonds join the fatty acids to the glycerol molecule (condensation reactions form them, hydrolysis breaks them/forms water).
-difference between triglycerides come from fatty acid variations.
Fatty Acids
-all fatty acids have a carboxyl group with a hydrocarbon chain extension.
-if the hydrocarbon chain has no carbon carbon double bonds, it is saturated.
-if the hydrocarbon chain has carbon carbon double bonds, it is unsaturated.
-one double carbon carbon bond is monounsaturated, multiple is polyunsaturated.
-difference between triglycerides come from fatty acid variations.
Fatty Acids
-all fatty acids have a carboxyl group with a hydrocarbon chain extension.
-if the hydrocarbon chain has no carbon carbon double bonds, it is saturated.
-if the hydrocarbon chain has carbon carbon double bonds, it is unsaturated.
-one double carbon carbon bond is monounsaturated, multiple is polyunsaturated.
Phospholipids
-they are like triglycerides, except one fatty acid is replaced my a phosphate group.
-it is a polar molecule (a hydrophilic head and a hydrophobic tail).
-it is a polar molecule (a hydrophilic head and a hydrophobic tail).
Test for Lipids
-this is known as the emulsion test.
1) in the test tube, add ethanol to the sample.
2) shake thoroughly to dissolve any lipid in the sample.
3) slowly pour the contents into a separate test tube of water.
4) observe a cloudy-white emulsion form near the surface (cloudy due to lipid refracting light).
5) to test this, you could repeat but with water rather than ethanol, observing that no cloud forms.
1) in the test tube, add ethanol to the sample.
2) shake thoroughly to dissolve any lipid in the sample.
3) slowly pour the contents into a separate test tube of water.
4) observe a cloudy-white emulsion form near the surface (cloudy due to lipid refracting light).
5) to test this, you could repeat but with water rather than ethanol, observing that no cloud forms.
