Carbon
-carbon likes to bond with carbon, forming a 'backbone'.
-organic molecules contain carbon.
-organic molecules contain carbon.
Organic Molecules
-many organic molecules (polymers) are made up of individual molecules (monomers).
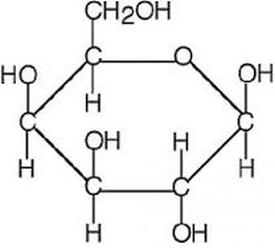
-a carbohydrate (polysaccharide) is made up from disaccharides (two monosaccharides).
-a carbohydrate (polysaccharide) is made up from disaccharides (two monosaccharides).
Monosaccharides
-soluble molecules (small enough to dissolve).
-they are all reducing sugars (as well as some disaccharides, excluding sucrose).
-have the formula (CH2O)n.
-glucose, fructose and galactose are hexose monosaccharides (have 6 carbon atoms per molecule), so C6H12O6.
-they contain the same elements, but are structures differently, giving slightly different sweet tastes.
-they are all reducing sugars (as well as some disaccharides, excluding sucrose).
-have the formula (CH2O)n.
-glucose, fructose and galactose are hexose monosaccharides (have 6 carbon atoms per molecule), so C6H12O6.
-they contain the same elements, but are structures differently, giving slightly different sweet tastes.
Test for Reducing Sugars
-reduction is the gain of electrons.
-reducing sugars can donate electrons to other chemicals (e.g. Benedict's reagent), to reduce it.
-there is a test you can do on a food sample to see if it contains reducing sugars.
1) add Benedict's reagent.
2) heat in a water bath at 90°C.
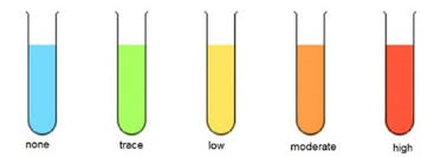
3) observe a colour change from blue to a orange-brown/brick red precipitate of copped oxide (the copper in the Benedict's gains electrons)
-reducing sugars can donate electrons to other chemicals (e.g. Benedict's reagent), to reduce it.
-there is a test you can do on a food sample to see if it contains reducing sugars.
1) add Benedict's reagent.
2) heat in a water bath at 90°C.
3) observe a colour change from blue to a orange-brown/brick red precipitate of copped oxide (the copper in the Benedict's gains electrons)
-this is a semi-quantitative test (it can be used to approximate the amount of reducing sugar).
-other than colour change, one could dry the solution to leave the copper oxide, then weigh this.
-it is hard to distinguish between very concentrated samples because no more Benedict's can be reduced into copper oxide after its run out.
-other than colour change, one could dry the solution to leave the copper oxide, then weigh this.
-it is hard to distinguish between very concentrated samples because no more Benedict's can be reduced into copper oxide after its run out.
Questions
1) Why do large molecules often contain carbon? (1 mark)
2) Why does Benedict's reagent turn brick red when heated with a reducing sugar? (3 marks)
2) Why does Benedict's reagent turn brick red when heated with a reducing sugar? (3 marks)